
Researchers Develop Tiny Human Retinas From Stem Cells In Lab Dish
Kiara Eldred, involved in growing tiny human retinas in a laboratory dish, considers the activity of nine-month-long scientific experiments sometimes to be like raising children.
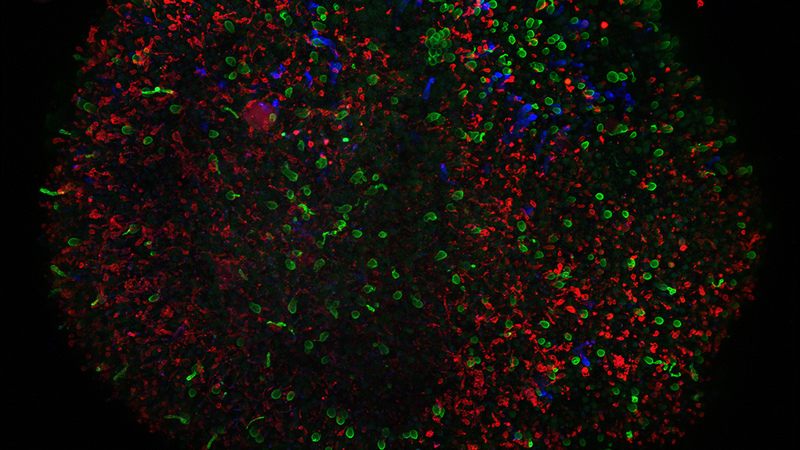
Eldred is a student pursuing graduation at Johns Hopkins University, begins by developing thousands of stem cells by feeding them all the required nutrients and chemicals eventually steering them to end up becoming the retina. The retina is the most crucial part of the eye which translates light into the signals that lead to vision. A painstaking exercise extending up to two weeks, the cells cultivate to typically generate 20 to 60 tiny balls of cells, called as retinal organoids. But as they start to mature, these retinas turn dirty and leave away lots of cells requiring them to be washed off every other day after they are fed. This process would go on at least for the first month and a half.
After continuous efforts for nine months with assiduous care, Eldred has got a batch of miniature human retinas that are able to respond to light, and the size is about two millimeters in diameter and have the shape that of tennis ball cut in half. But the process of growing the organoids is just only the first step.

But all these studies being taken up by Eldred and colleagues by using this system is to understand a very fundamental question about vision that is still a mysterious one with out any answer to it: How does color vision develop in human retinas?
The researchers were able to find it out that the blue cone cells develop first and these are responsible for detecting blue colored light and after this develops red and green photoreceptor cells. They are were able to find it out that thyroid hormone seems to be most critical signal that determines which light-detecting cells develop.
At the end, the researchers working on this project hope that those insights and studies may help in developing treatment for diseases because of which these light-detecting cells get depleted, like the macular degeneration. More deep understanding of the process may help to develop therapies for vision defects that develop in premature infants.
“The ideal goal would be to take a person’s cells, convert them into stem cells, and then reprogram them and put them back in the person and treat whatever the disease is,” said Robert Johnston Jr., a developmental neurobiologist at Johns Hopkins who heads the lab where Eldred works.
But the developed organoid has got their own limitations. In reality, Human retinas are approximately 10 times bigger that those organoids, Eldred said. Many questions remain to be answered about how well they mirror eye development in the fetus, as they don’t have many other peripheral structures.
“In the past, if we wanted to work out the developmental mechanisms underlying a particular process, we would turn to model organisms like mice or zebrafish,” said Thomas Reh, a scientist at the University of Washington who is involved in studying the development of the eye.
You May Also Read: Lockheed Martin Using Microsoft’s HoloLens To Build Spacecraft Faster For NASA